How To Draw Resonance Structures
How to Draw Resonance Structures, Rules, Examples, Bug
There are rules to follow drawing resonance structures pace past step. For molecules and ions, nosotros can draw several resonance structures and their stability is dissimilar from one structure to some other structure and yous should have the ability to identify stability of each structure.
Rules to draw resonance structures
- Locations of nuclei of atoms should not exist changed with structures. Structures are changed only with their electrons configuration.
- Number of total electron pairs should be same in every construction. Full electron pairs can be simplified as bonds and alone pairs.
Otherwise we can say, number of electron pairs = σ bonds + π bonds + lone pairs. If unpaired electrons exist, they besides should be same number in every molecules. (example NO2). - Energy of dissimilar structures should be aforementioned or close.
- Higher electronegative atoms have more take chances to hold negative charges to obtain more stability to the molecule or ion. Every bit an instance, when sulfur and oxygen is in the molecule, oxygen has the college ability to hold the negative charge (Southward2Oiii ii-).
Guidelines to follow
To draw all resonance structures, take the lewis structure we drawn past using VESPR rule.
In resonance structures, information technology does not require to evidence transformation of electrons by arrows. Just, to identify each resonance structures, it is practiced to show arrows. In post-obit examples, arrows are used to show electrons transformation.
Next, we will learn how to employ those rules to describe resonance structures properly. Lets draw resonance structures of nitrate ion.
Resonance structures of nitrite ion (NOthree -)
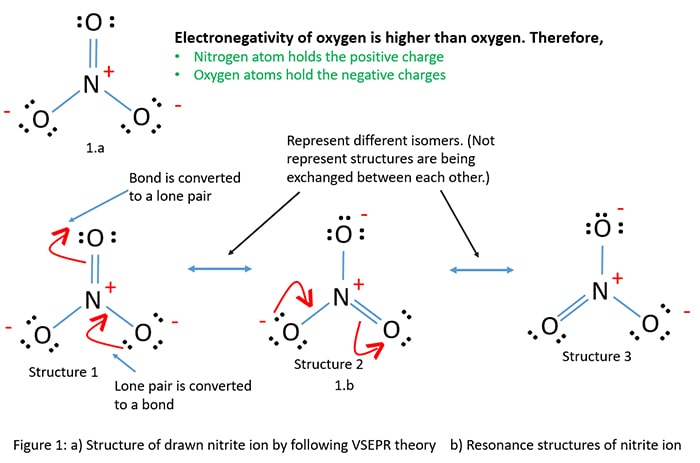
Nitrogen atom has the greatest possibility to exist the middle cantlet than oxygen atom co-ordinate to theory of an atom which has a greater valence should be the centre atom. Therefore, three oxygen atoms are located around the nitrogen atom. When nosotros draw the construction of the nitrite ion past following "VSEPR rule". Then it is as the figure 1.a .
Go along octal of oxygen and nitrogen atoms
Remember that, octal of oxygen and nitrogen atoms cannot be exceeded because both oxygen and nitrogen atoms are located in the second flow and have just s and p orbitals.
In the nitrite ion, there is a -1 charge. According to the dominion number 4, an oxygen atoms should keep negative charges considering electronegativity of oxygen is college than nitrogen. In NOiii -, there are two -1 charges on 2 oxygen atoms and +i charge on nitrogen atom.
Now we have ane structure. Now we endeavor to depict more than structures past changing the bonds and lone pairs keeping locations of atoms without irresolute (rule number 1).
Bond to lone pair, lone pair to bond
A lonely pair is converted to a bail. With that, a bond should be converted to a alone pair by keeping the octal of nitrogen and oxygen atoms. So it follows rule number 2 which says number of total electron pairs should be same in every structure.
With conversion of a bail to a alone pair and lone pair to a bail, double bail becomes a single bail and unmarried bond become double bail respectively. Study the resonance structures of NO2 - ion (Figure 1.b)
Iii resonance structures
3 resonance structures tin can be drawn. In all resonance structures (mutual for all drawn resonance structures), two oxygen atoms have -1 charge and nitrogen atom has a +ane charge.
Stability of NO3 - resonance structures
You encounter, all fatigued three resonance structures are like because,
- There are 1 double bond between nitrogen and oxygen atom and ii unmarried bonds nitrogen and oxygen atoms.
- Always at that place are -1 charges on two oxygen atoms and +1 charge on nitrogen atom.
Therefore all three resonance structures have equal stability. In next examples, yous may see, different isomers' stabilities for another molecules are not same as NOiii - .
Sulfur dioxide (SO2) molecule resonance structures
 molecule resonance structures.jpg)
We can describe three resonance structures for Then2 molecule. You can see first 2 structures take charges on atoms. But in third construction, at that place are no charges on atoms. So nosotros can understand, construction iii is more stable than other ii construction.
Nitrogen dioxide (NOii) molecule
 resonance structures.jpg)
Nitrogen dioxide as well testify two resonance structures. Merely, their stability is same.
How do you lot employ the dominion, negatively charges should be on most electronegative atoms in this ii resonance structures?
You see in both resonance structures, nosotros have marked the negative charge on oxygen atom and positive charge on nitrogen atom.
Resonance structures nitrous oxide (NorthwardiiO) molecule
 resonance structures.jpg)
We can draw three resonance structures for Northward2O.
Nitrite ion (NO2 -)
Nitrite ion is a -1 charge. So that negative charge should be kept on oxygen atom. Again we see, in most stable structures negative charges should be put on oxygen atoms.
 resonance structures.jpg)
Related Tutorials to resonance structures
Source: https://www.chemistryscl.com/general/resonance-structures-examples/index.php
Posted by: parkblegame94.blogspot.com


0 Response to "How To Draw Resonance Structures"
Post a Comment